A comparison between intermittent self-catheterization and transurethral indwelling catheterization in patients who underwent radical hysterectomy for cervical cancer: a systematic review
Introduction
Cervical cancer is one of the most common cancers in women and leads to relatively high morbidity and mortality. Most patients with cervical cancer require radical hysterectomy (1). During this surgery, the uterus and cervix may be removed; at the same time, the supporting tissues, upper part of the vagina, and nerves in the operative area are also removed, leading to postoperative bladder dysfunction (2,3). Even nerve-sparing radical hysterectomy causes damage to the nerves supplying the bladder (4). Thus, patients are left with great discomfort and diminished quality of life. Postoperative catheterization is performed for management of urinary retention, incontinence, dysuria, and sensory loss.
Urinary catheters are inserted suprapubically or via the urethra to help urine to pass, tissue to heal, and to assess voiding function. There has been no consensus on the optimal method of catheterization in patients undergoing hysterectomy for cervical cancer, and the choice is still guided by the individual surgeon’s experience and anecdotal evidence (5,6). Since suprapubic catheterization is invasive and may be accompanied by visceral injury or bleeding (7), this systematic review focused on a comparison between intermittent self-catheterization (ISC) and transurethral indwelling catheterization (TIC) after hysterectomy for cervical cancer.
An indwelling catheter is left in place after surgery. ISC can be used to replace an indwelling catheter within a week, when most patients still have voiding dysfunction, with training in self-catheterization until voiding function recovery (VFR). TIC can be used to replace a postoperative indwelling catheter after a relatively longer time, when most patients have already recovered from voiding dysfunction; the remaining patients with voiding dysfunction will undergo repeat catheterization as determined by the surgeon. We defined the time until first postoperative indwelling catheter removal (FPICR time) as the duration from the first postoperative day to the day when the catheter was removed for the first time to assess voiding function. In recent years, ISC has been increasingly accepted by urological and gynecological departments (8,9). Many studies have shown that patients prefer ISC because of improved quality of life (6,10,11). In addition, patients can manage ISC without difficulty after professional training (11,12). However, TIC, as the conventional method, is still widely used in gynecological wards.
This review aimed to determine which transurethral catheterization method is better for patients undergoing radical hysterectomy for cervical cancer. A shorter VFR time would lead to a lower rate of urinary tract infection (13), less discomfort, higher quality of life, and less cost (6,10,11). Therefore, we chose VFR time as the main outcome. In this review, the time to VFR was defined as the time of final catheter removal (the duration from the first postoperative day to the day when the catheter was removed, according to the criteria for VFR (Table 1).
Table 1
| Author, year | Location | Time | Methods | FIGO stage | Operation type | Criteria for VFR |
|---|---|---|---|---|---|---|
| Raspagliesi |
Italy | January 2000 to December 2002 | Case series | IB1, IB2*, IIA*, IIB* | NS Piver III RH with systematic PL | PVR <100 mL using 10-Fr catheter |
| Roberts & Naik ( |
UK | July 1, 1999 to June 31, 2002 | RCT | IB1 | RH | PVR <100 mL using catheter |
| Maneschi |
Italy | June 2008 to December 2009 | Case series | IB1, IB2*, IIB* | Class III to IV RH with lymphadenectomy | PVR <50 mL** |
| Oh, Park, & Oh ( |
Korea | 2004 to 2008 | Retrospective cohort study | IB–IIB | RH with PL | PVR <100 mL** |
| Bogani |
Italy | May 2004 to December 2012 | Prospective cohort study | IA, IB1, IB2*, IIA*, IIB* | Types II and III LRH with systematic PL, some cases with para-aortic lymphadenectomy | Not mentioned |
| Hao |
China | June 2011 to June 2013 | RCT | IB1, IIA1 | NS Piver III LRH with PL | PVR <100 mL assessed by ultrasound |
| Gong |
China | February 2012 to April 2015 | RCT | IB1, IB2*, IIA*, IIB* | type C RH | PVR <200 mL assessed with 3-dimensional ultrasound 48 h after catheter removal |
*, neoadjuvant chemotherapy or chemo-radiation treatment was performed; **, the method for assessing PVR was not mentioned. VFR, voiding function recovery; NS, nerve-sparing; RH, radical hysterectomy; PL, pelvic lymphadenectomy; PVR, post-void residual; RCT, randomized controlled trial.
Methods
We used the terms “cervical cancer (MeSH) AND ((indwelling urethral catheterization (MeSH)) OR (intermittent self-catheterization (MeSH)))” to search PubMed, the Cochrane Library, and Embase from inception to July 2017.
All trials involving the application of ISC or TIC after radical hysterectomy for cervical cancer were included. The inclusion criteria were as follows: (I) clinical stages of cervical cancer from IA to IIB; (II) use of either laparoscopic or open hysterectomy; (III) catheter removal time as a reported outcome; and (IV) any type of study design, including randomized controlled trials (RCTs), cohort studies, and case series.
The exclusion criteria were as follows: (I) non-human research, reviews, case reports and letters; (II) duplicated studies or data from the same hospital during the same period; (III) research data that did not mention catheter removal time; and (IV) studies that included patients with diseases other than cervical cancer.
Two reviewers independently screened the articles for data extraction, and their findings were cross-checked. We extracted the author, year, location, study time and methods, sample size, International Federation of Gynecology and Obstetrics (FIGO) stage, operation type, sample size, FPICR time, criteria for VFR, VFR time, and the difference between VFR and FPICR time. Summaries are presented in Tables 1 and 2.
Table 2
| Author, year | Sample size of every group in each study | FPICR time (days) | VFR time (days) | VFR (%) on a specific postoperative day | Quality score |
|---|---|---|---|---|---|
| Raspagliesi |
ISC: n=23 | 3 | 6.5 (SD, 4.9) | 4th day: 39%; 5th day: 43% | 7 |
| Roberts & Naik ( |
ISC: n=19 | 4 | 17 (range, 7–90) | – | 9 |
| Maneschi |
ISC: n=15 | 3 | 8 (range, 6–28) | 10th day: 80% | 7 |
| Oh, Park, & Oh ( |
ISC: n1=42; TIC: n2 (one surgeon) =114, n3 (other surgeons) =29 | 8 | n1: 8.3 (SD, 1.1)*, n2: 13.1 (SD, 3.3), n3: 13.0 (SD, 1.1) | – | 7 |
| Bogani |
TIC: n1 (NS-LRH) =33, n2 (CLRH) =63 | 2 | n1: 3.5 (range, 2–7), n2: 5.5 (range, 4–7) | – | 8 |
| Hao |
TIC: n1 (CUSA) =24, n2 (other techniques) =21 | 7 | n1: 7.13 (SD, 0.61), n2: 9.00 (SD, 3.24) | – | 7 |
| Gong |
TIC: n1 (NC-BRC) =128, n2 (C-BRC) =70 | 13 | n1: 14.78 (SD, 7.74), n2: 14.83 (SD, 9.91) | 14th day: 85.5% | 9 |
*, no follow-up of self-catheterization. FPICR, first postoperative indwelling catheter removal; VFR, voiding function recovery; ISC, intermittent self-catheterization; TIC, transurethral indwelling catheterization; SD, standard deviation; NS, nerve-sparing; LRH, laparoscopic radical hysterectomy; CLRH, conventional laparoscopic radical hysterectomy; CUSA, Cavitron Ultrasonic Surgical Aspirator; NC-BRC, non-clamping 2 days before removal of catheter; C-BRC, clamping 2 days before removal of catheter.
The quality of RCTs, cohort studies, and case series was assessed by 2 independent reviewers using the Joanna Briggs Institute Meta-Analysis of Statistics Assessment and Review Instrument (JBI-MAStARI) RCT quality evaluation list, Newcastle-Ottawa cohort study quality evaluation list, and the JBI-MAStARI descriptive/case series quality evaluation list, respectively. Referring to a prior published study (20), we defined high quality as a score >7, medium quality as a score of 5–7, and low quality as a score <5. The quality assessment results for the included studies are reported in Table 2.
Results
Search results
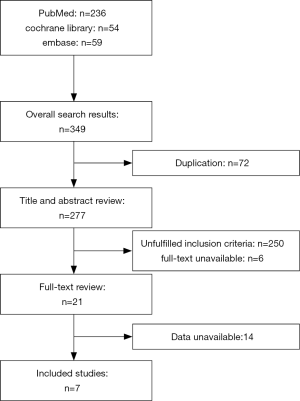
The total number of included patients was 581, ranging from 15 to 128 cases in each study. We first obtained 349 studies from a search of PubMed (n=236), the Cochrane Library (n=54), and Embase (n=59). After excluding duplications (n=72), studies that did not fulfill the inclusion criteria (n=250) as judged by the titles and abstracts, and studies for which the full text was unavailable (n=6), 21 studies remained. Finally, 7 studies met all the inclusion criteria and were reviewed in this study. The flow chart of literature search is presented in Figure 1.
Basic characteristics
Overall, 3 studies included samples of ISC only (6,14,15), 3 reported TIC only (17-19), and 1 included samples that compared ISC to TIC (16). The FIGO stages of cervical cancer ranged mainly from IB to IIB (Table 1). Only 1 study recruited a small number of IA FIGO stage patients (16-19). Criteria for VFR varied from post-void residual (PVR) urine volume <50 mL to PVR <200 mL among the included studies. We found that PVR <100 mL was the most accepted criterion for VFR (Table 1). The PVR was assessed either with a catheter or through ultrasound examination. Three of the 7 studies included samples from Italy, 2 were from China, 1 was from the UK, and 1 was from Korea. Quality assessment resulted in 3 high-quality studies and 4 medium-quality studies.
Results of VFR
For ISC, the reported VFR time ranged widely from 6.5 to 17 days (6,14,15); for TIC, the VFR time ranged from 3.5 to 14.8 days (16-19). The results for ISC and TIC were both too variable to permit even a general impression on the VFR time after radical hysterectomy for cervical cancer. The results are presented in Table 2.
The time to FPICR and assessment of PVR also showed a wide range of results, with 3 to 8 days for ISC and 7 to 14 days for TIC.
As seen from the data, 39%, 43%, 80%, and 85.5% of patients reached VFR on the fourth, fifth, tenth, and fourteenth postoperative day, respectively (14,15,19). These data show that the percentage of patients with VFR increased over time.
Of note, the VFR time in Italy was only 3.5 to 8 days and the FPICR time was only 3 days (15,20), while both times in the studies from other countries were relatively longer.
Discussion
Two other systematic reviews attempted to determine which catheterization method is better but arrived at no conclusion (21,22). They examined broad categories such as urogenital, abdominal, and cardiac surgeries. The studies also compared length of hospital stay, infection rates, VFR time, cost, and satisfaction rates, among others. Consideration of multiple factors in combination yielded too many variables to enable a conclusion. To focus on a specific area and a primary factor for assessment of superiority between 2 catheterization methods, we limited our research scope to only radical hysterectomy for cervical cancer and VFR time. However, the VFR time range was too wide to obtain a single value for either ISC or TIC. Although the duration of VFR seemed slightly longer for ISC than for TIC (6.5 to 17 days versus 3.5 to 14. 8 days), we could not conclude that TIC was better for VFR time than ISC from such a small difference.
We initially thought that the major reason for the wide range in VFR time was the extent of the procedure. The various clinical stages required different approaches to radical hysterectomy. The difference in surgical extent resulted in different influences on postoperative bladder function. This would cause bias in a comparison of the 2 catheterization methods and prevent a firm conclusion. Second, the PVR used for catheter removal ranged from <50 to 200 mL in this review. This wide range led to significant bias in the comparison, which then yielded inconclusive results.
Although the percentage of patients who reached VFR increased with postoperative time, the results indicated that delayed removal of the indwelling catheter and assessment of voiding function could increase infection risk and diminish quality of life (22,23). Some studies supported early removal of the postoperative indwelling catheter to prevent bladder dysfunction (5,24,25). However, our findings showed a wide range in the FPICR time for assessment of PVR after radical hysterectomy for cervical cancer.
The VFR time in Italy was shorter than in other countries. Surgeons and nurses in Italy may have used a more optimized strategy of catheterization. One detailed description of catheterization was documented in an Italian study. The postoperative indwelling Foley catheter was only used for 3 days, and was repeatedly closed for 3 h and opened for 15 min on the second and third days; on the fourth and fifth days, the bladder was drained every 3 h with a 10-Fr catheter until PVR was <100 mL; patients went home with ISC if VFR had not been reached (14).
There were limitations in this systematic review. First, the included studies were not all RCTs and the number of samples in some included studies was small. Second, the surgical types, surgical techniques, and criteria for catheter removal could also affect the VFR time (26), but these factors were not used for subgroup analysis due to lack of sufficient data. With all these limitations, the conclusion must be regarded with caution.
We concluded that the results were insufficient to enable a comparison between ISC and TIC. Therefore, we failed to identify which catheterization method was better for VFR in patients with cervical cancer who have undergone radical hysterectomy. High-quality studies are still needed.
Because of the wide range in FPICR times, VFR times, and PVR criteria for catheter removal, different types of radical hysterectomy may require different guidelines for catheterization. A worldwide effort to standardize catheterization after radical hysterectomy for cervical cancer is also recommended.
Acknowledgments
We would like to thank Wiley Editing Services for language editing assistance, all researchers of the included original studies, and participants related to these studies.
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://gpm.amegroups.org/article/view/10.21037/gpm.2019.02.01/coif). ZYL serves as an unpaid editorial board member of Gynecology and Pelvic Medicine from Jun 2018 to May 2020. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Hellebrekers BW, Zwinderman AH, Kenter GG, et al. Surgically-treated early cervical cancer: Prognostic factors and the significance of depth of tumor invasion. Int J Gynecol Cancer 1999;9:212-9. [Crossref] [PubMed]
- Raspagliesi F, Ditto A, Fontanelli R, et al. Type II versus Type III nerve-sparing radical hysterectomy: comparison of lower urinary tract dysfunctions. Gynecol Oncol 2006;102:256-62. [Crossref] [PubMed]
- Sevin BU, Nadji M, Lampe B, et al. Prognostic factors of early stage cervical cancer treated by radical hysterectomy. Cancer 1995;76:1978-86. [Crossref] [PubMed]
- Trimbos JB, Maas CP, Deruiter MC, et al. A nerve-sparing radical hysterectomy: guidelines and feasibility in Western patients. Int J Gynecol Cancer 2001;11:180-6. [Crossref] [PubMed]
- Turnbull H, Burbos N, Abu-Freij M, et al. A novel approach to postoperative bladder care in women after radical hysterectomy. Arch Gynecol Obstet 2012;286:1007-10. [Crossref] [PubMed]
- Roberts K, Naik R. Catheterization options following radical surgery for cervical cancer. Br J Nurs 2006;15:1038-44. [Crossref] [PubMed]
- Parikh A, Chapple CR, Hampson SJ. Suprapubic catheterisation and bowel injury. Br J Urol 1992;70:212-3. [Crossref] [PubMed]
- Anderson JB, Grant JB. Postoperative retention of urine: a prospective urodynamic study. BMJ 1991;302:894-6. [Crossref] [PubMed]
- Kleeman S, Goldwasser S, Vassallo B, et al. Predicting postoperative voiding efficiency after operation for incontinence and prolapse. Am J Obstet Gynecol 2002;187:49-52. [Crossref] [PubMed]
- Cobussen-Boekhorst H, Hermeling E, Heesakkers J, et al. Patients' experience with intermittent catheterisation in everyday life. J Clin Nurs 2016;25:1253-61. [Crossref] [PubMed]
- Pilloni S, Krhut J, Mair D, et al. Intermittent catheterisation in older people: a valuable alternative to an indwelling catheter? Age Ageing 2005;34:57-60. [Crossref] [PubMed]
- Edwards M, Borzyskowski M, Cox A, et al. Neuropathic bladder and intermittent catheterization: social and psychological impact on children and adolescents. Dev Med Child Neurol 2004;46:168-77. [Crossref] [PubMed]
- Hakvoort RA, Thijs SD, Bouwmeester FW, et al. Comparing clean intermittent catheterisation and transurethral indwelling catheterisation for incomplete voiding after vaginal prolapse surgery: a multicentre randomised trial. BJOG 2011;118:1055-60. [Crossref] [PubMed]
- Raspagliesi F, Ditto A, Fontanelli R, et al. Nerve-sparing radical hysterectomy: a surgical technique for preserving the autonomic hypogastric nerve. Gynecol Oncol 2004;93:307-14. [Crossref] [PubMed]
- Maneschi F, Ianiri P, Sarno M, et al. Nerve-sparing class III-IV radical hysterectomy: urodynamic study and surgical technique. Int J Gynecol Cancer 2012;22:675-80. [Crossref] [PubMed]
- Oh JK, Park NH, Oh SJ. Effect of the systematised critical pathway protocol on emptying failure as a secondary complication of radical hysterectomy due to uterine cervix cancer. J Clin Nurs 2014;23:1702-7. [Crossref] [PubMed]
- Bogani G, Cromi A, Uccella S, et al. Nerve-sparing versus conventional laparoscopic radical hysterectomy: a minimum 12 months' follow-up study. Int J Gynecol Cancer 2014;24:787-93. [Crossref] [PubMed]
- Hao M, Wang Z, Wei F, et al. Cavitron Ultrasonic Surgical Aspirator in Laparoscopic Nerve-Sparing Radical Hysterectomy: A Pilot Study. Int J Gynecol Cancer 2016;26:594-9. [Crossref] [PubMed]
- Gong Y, Zhao L, Wang L, et al. The effect of clamping the indwelling urinary catheter before removal in cervical cancer patients after radical hysterectomy. J Clin Nurs 2017;26:1131-6. [Crossref] [PubMed]
- Zheng J, Feng X, Yang Z, et al. The comprehensive therapeutic effects of rectal surgery are better in laparoscopy: a systematic review and meta-analysis. Oncotarget 2017;8:12717-29. [PubMed]
- Kidd EA, Stewart F, Kassis NC, et al. Urethral (indwelling or intermittent) or suprapubic routes for short-term catheterisation in hospitalised adults. Cochrane Database Syst Rev 2015;CD004203 [PubMed]
- Phipps S, Lim YN, McClinton S, et al. Short term urinary catheter policies following urogenital surgery in adults. Cochrane Database Syst Rev 2006;CD004374 [PubMed]
- Naik R, Maughan K, Nordin A, et al. A prospective randomised controlled trial of intermittent self-catheterisation vs. supra-pubic catheterisation for post-operative bladder care following radical hysterectomy. Gynecol Oncol 2005;99:437-42. [Crossref] [PubMed]
- Chamberlain DH, Hopkins MP, Roberts JA, et al. The effects of early removal of indwelling urinary catheter after radical hysterectomy. Gynecol Oncol 1991;43:98-102. [Crossref] [PubMed]
- Campbell P, Casement M, Addley S, et al. Early catheter removal following laparoscopic radical hysterectomy for cervical cancer: assessment of a new bladder care protocol. J Obstet Gynaecol 2017;37:970-2. [Crossref] [PubMed]
- Cibula D, Slama J, Velechovska P, et al. Factors affecting spontaneous voiding recovery after radical hysterectomy. Int J Gynecol Cancer 2010;20:685-90. [Crossref] [PubMed]
Cite this article as: Ming X, Wang X, Li ZY. A comparison between intermittent self-catheterization and transurethral indwelling catheterization in patients who underwent radical hysterectomy for cervical cancer: a systematic review. Gynecol Pelvic Med 2019;2:5.