Laparoscopic management of ureteral endometriosis associated with hydronephrosis
Introduction
Ureteral endometriosis (UE) is an important gynecologic clinical disorder characterized by the ectopic presence and growth of functional endometrial tissue, glands, and stroma, outside the uterus, and is clinically associated with pelvic pain and infertility. Urinary tract endometriosis occurs in 1–5.5% of women with endometriosis; it involves the bladder in 70–85% of cases and the ureter in 9–23% of the cases (1). Although severe ureteral obstruction may lead to hydronephrosis and renal failure, it is rather difficult to diagnose UE because of the frequent lack of signs or symptoms and the absence of physical findings pathognomonic of this condition. The surgical treatment of UE aims to relieve ureteral obstruction and avoid disease recurrence. It includes conservative ureterolysis or radical approaches like ureterectomy with end-to-end anastomosis or ureteroneocystostomy performed in relation to the type of ureteral involvement (2). Finding the optimal diagnostic and therapeutic management for UE is difficult owing to the fact that most studies enroll a small number of patients, are uncontrolled and not randomized, and have short-term follow-up.
In the case reported here, a 23-year-old unmarried woman with no history of sexual intercourse was admitted to the hospital due to menstrual changes for 4 months, left ovarian cyst, and right hydronephrosis for 1 month. Clinical examination results are described below.
Anal examination: the vulva was normally developed and the rectal mucosa was smooth, while the uterus was in a horizontal position, evenly enlarged to the size of over 2 months of pregnancy, slightly tender, and a little hard. A tumor, poorly movable, with a diameter of 5 cm could be found in the left adnexa. Bilateral uterosacral and cardinal ligaments were not thickened or tender.
Pelvic and urinary ultrasound: there was a left ovarian cyst, adenomyosis, and right-sided hydronephrosis. The right upper ureter was distorted, and the proximal lumens were dilated, of which the wider part was about 10 mm wide.
Computed tomography urography (CTU): the right kidney and ureter were dilated with the occurrence of hydronephrosis. The right distal ureter was narrowed and could not be clearly seen.
Magnetic resonance imaging (MRI): there was endometrioma of the left ovary. Adenomyosis could be seen invading and surrounding the pelvic segment of the right ureter resulting secondarily in right ureter, renal pelvis, calyces that were significantly dilated. The renal parenchyma was thinned under slight compression.
Finally, she was diagnosed with (I) endometrioma of the left ovary, (II) right ureteral obstruction, (III) and adenomyosis; she was treated by right ureter double-J stent insertion plus laparoscopic right ureter dissociation plus laparoscopic deep infiltrating endometriosis (DIE) resection (Figure 1). GnRH-a was applied for 4 months after surgery and double-J tube was withdrawn 2 months later. CT examination suggested the hydronephrosis had not been improved.

Surgical technique
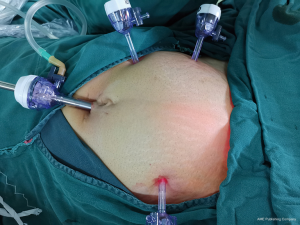
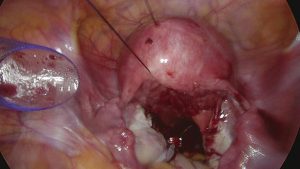
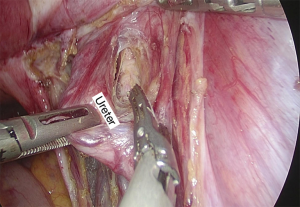
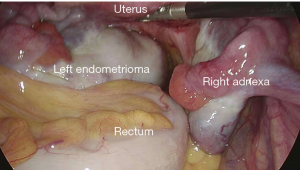
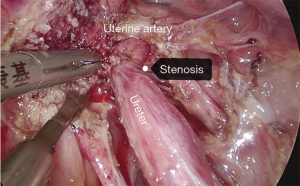
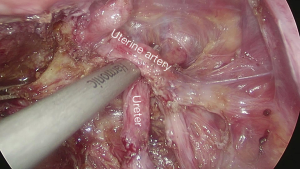
Under general anesthesia with intubation, the patient underwent the surgery in a cystolithotomy position with the buttocks higher than the head, with standard technique including the use of 10-mm operative laparoscope and three 5-mm ancillary trocars in the presence of 12-mmHg intra-abdominal pressure. The surgical incision was selected as shown in Figure 2. Before laparoscopic surgery, the right ureteral double-J tube implantation was performed under cystoscope with slight resistance but was nonetheless successful. As the patient had no history of sexual intercourse, the uterus could not be lifted through the vagina. The uterus was suspended to expose the surgical field as shown in Figure 3. The surgical exploration of the pelvic and abdominal cavity revealed that the uterus was full, mobile, and slightly enlarged. The left ovarian cyst was 5 cm in diameter, no abnormalities were found in the right adnexa, and a mild endometriosis lesion was visible in the peritoneum of the Douglas cul-de-sac as shown in Figure 4. After the ectopic cyst of the left ovary was removed, the right parietal peritoneum was opened to expose and dissociate the right ureter as shown in Figure 5. In the uterine artery, the ureter was indistinctly demarcated from the surrounding tissue resulting in stenosis due to compression, as shown in Figure 6. The ectopic lesion tissue around the urinary duct was dissociated and removed fully and carefully so as not to damage the uterine artery, as shown in Figure 7. The peritoneal was then closed.
Comments
Depending on the location of the UE lesion, the condition can be divided into two categories (4-6). The first is extrinsic endometriosis accounting for about 80% of cases, most of which are caused by ectopic lesions involving the cardinal-uterosacral ligament and retroperitoneum spreading to the ureter. These lesions infiltrate the peripheral tissues, causing fibrosis and consequent narrowing of the ureter, eventually leading to ureteral obstruction and hydronephrosis. The second is intrinsic endometriosis, accounting for about 20% of cases, most of which are characterized by fibrous thickening of the ureteral wall, submucosal hyperplasia, or polypoid neoplasms that protrude from the mucosal layer penetrating the lumen. Surgical treatment should be considered as the first option for ureteral obstruction caused by UE. The principle of surgery is to remove the obstruction, eliminate symptoms, and preserve kidney function (7). The main surgical management includes conservative ureterolysis or radical approaches like ureterectomy with end-to-end anastomosis, ureteroneocystostomy, or nephroureterectomy (2). The surgical treatment depends on the extension of UE and on renal function.
There is no current consensus as to which surgical procedure is optimal for this condition, as most clinical doctors rely on personal experience and technology which contributes to two common problems: (I) it is difficult to achieve the desired therapeutic effect because of the incomplete removal of the lesion resulting from worries about injury to the ureter and bladder, which is a potential trigger for recurrence; (II) partial removal of the involved bladder, ureter or kidney, or removal of the organ causes unnecessary damage to the patient. Because the incidence of the extrinsic cases is much higher than that of the intrinsic ones, most scholars recommend the preferred surgical approach for release of ureteral adhesions (8). However, other scholars believe that ureterolysis is only indicated for patients with endometriosis with mild hydronephrosis and without ureteral obstruction as diagnosed by imaging examination. For patients with moderate-to-severe ureteral obstruction, it is recommended to remove the involved ureteral stricture and perform end-to-end anastomosis or ureteroneocystostomy to avoid further damage of renal function (9). For the subgroup of women with moderate ureteral hydronephrosis on preoperative imaging, for which the current surgical results are still controversial, we suggest well powered, randomized controlled trials (RCTs) be completed in order to more accurately investigate the optimal management and outcomes.
Our experience is that if the double-J stent can be successfully placed before surgery, ureterolysis is preferable to the partial removal of the ureter. Meanwhile, the so-called release of adhesions can be done to not only completely detach the ureter from the pelvic wall, but also to thoroughly remove the lesions and scar tissues around the ureter. From the perspective of surgical difficulty, the release of severe periureteral compression requires higher surgical skills and rich clinical experience without damaging the ureter.
In our case, the double-J stent was successfully placed preoperatively, and the intraoperative exploration showed serious periureteral fibrosis, obviously compressing the ureter. The surrounding ectopic lesions were fully excised, the ureter was dissociated, and GnRH-a was applied postoperatively. CT reexamination 3 months after the operation showed the hydronephrosis remained unsatisfactorily improved. This case proves that it is not advisable to choose the surgical method for UE relying on personal experience alone. The appropriate surgical treatment depends on more detailed clinical examination, the individual patient condition, lesion location, lesion degree and technical characteristics. A close follow-up with regularly scheduled clinical evaluation and imaging should also be arranged in order to compare the relative risks of persistent and recurrent disease of the surgical options available.
Acknowledgments
The video was awarded the second prize in the First International Elite Gynecologic Surgery Competition (2019 Masters of Gynecologic Surgery).
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Gynecology and Pelvic Medicine for the series “Award-Winning Videos from the First International Elite Gynecologic Surgery Competition (2019 Masters of Gynecologic Surgery)”. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://gpm.amegroups.org/article/view/10.21037/gpm.2019.12.03/coif). The series “Award-Winning Videos from the First International Elite Gynecologic Surgery Competition (2019 Masters of Gynecologic Surgery)” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Berlanda N, Vercellini P, Carmignani L, et al. Ureteral and vesical endometriosis. Two different clinical entities sharing the same pathogenesis. Obstet Gynecol Surv 2009;64:830-42. [Crossref] [PubMed]
- Barra F, Scala C, Biscaldi E, et al. Ureteral endometriosis: a systematic review of epidemiology, pathogenesis, diagnosis, treatment, risk of malignant transformation and fertility. Hum Reprod Update 2018;24:710-30. [Crossref] [PubMed]
- Li Y, Wang Y, Huang Y, et al. Right ureter double-J stent insertion + laparoscopic right ureter dissociation + laparoscopic DIE resection. Asvide 2019;6:352. Available online: http://www.asvide.com/watch/33037
- Camanni M, Delpiano EM, Bonino L, et al. Laparoscopic conservative management of ureteral endometriosis. Curr Opin Obstet Gynecol 2010;22:309-14. [Crossref] [PubMed]
- Camanni M, Bonino L, Delpiano EM, et al. Laparoscopic conservative management of ureteral endometriosis: a survey of eighty patients submitted to ureterolysis. Reprod Biol Endocrinol 2009;7:109. [Crossref] [PubMed]
- Palla VV, Karaolanis G, Katafigiotis I, et al. Ureteral endometriosis: A systematic literature review. Indian J Urol 2017;33:276-82. [Crossref] [PubMed]
- Huang JZ, Guo HL, Li JB, et al. Management of ureteral endometriosis with hydronephrosis: Experience from a tertiary medical center. J Obstet Gynaecol Res 2017;43:1555-62. [Crossref] [PubMed]
- Darwish B, Stochino-Loi E, Pasquier G, et al. Surgical Outcomes of Urinary Tract Deep Infiltrating Endometriosis. J Minim Invasive Gynecol 2017;24:998-1006. [Crossref] [PubMed]
- Gobara M, Akino H, Nishibuchi S, et al. Genitourinary endometriosis: report of 2 cases and clinical aspects of patients in Japan. Hinyokika Kiyo 1994;40:349-52. [PubMed]
Cite this article as: Li Y, Wang Y, Huang Y, An L, Feng J, Qi L. Laparoscopic management of ureteral endometriosis associated with hydronephrosis. Gynecol Pelvic Med 2019;2:28.