
Interval debulking surgery for advanced ovarian cancer: when, how and why?
Introduction
Ovarian cancer is currently the seventh most common cancer in women and the leading cancer-related cause of gynecological mortality in developed countries. Furthermore, ovarian cancer is diagnosed at an advanced stage in most cases (75%) (1) and its symptoms are often vague and underestimated by patients.
Because of this, the five-year European survival rate after diagnosis is about 30–35% (2). However, while in the early stages (FIGO stage I–IIa) surgery may be sufficient to cure the majority of patients and to establish those who might benefit from adjuvant therapy, in the advanced stages (FIGO stage III–IVb) in which the disease has spread beyond the pelvis, a combination of surgery with cytoreduction effort and chemotherapy are necessary to obtain the best prognosis (3).
The standard treatment of advanced ovarian cancer (FIGO stage III–IV) currently consists of a primary debulking surgery (PDS) via open surgery aimed to achieve the complete resection of disease followed by adjuvant platinum-based chemotherapy (4). Following the results of the study by Griffiths et al. of 1975, a correlation between overall survival (OS) and the size of residual tumor after PDS was already known, and actually the tumor residue represents the most significant prognostic factor, together with other independent factors such as: age, performance status, histological grade, FIGO stage and histotype (5,6).
Because of the importance of the residual tumor after PDS, the definition of what makes debulking surgery “optimal” has changed over time. In the past years the optimum was considered to achieve when residual disease was no more neoplastic implants <2 cm of maximum diameter; instead today the goal is to reach no macroscopically detectable disease at the end of the surgery, the so-called TR0 (7). Indeed, patients who achieved TR0 have been shown to have better survival than those with residual disease <1 cm (optimal cytoreduction) and >1 cm (suboptimal cytoreduction) (6,8).
The feasibility of complete cytoreduction depends on the resectability of the tumor and the operability of patients, respectively related to the burden of disease and patients’ comorbidities, which might influence the tolerability of an extensive radical surgery. Of note, no less important, are the skills of the surgical team (9-12).
When PDS is not an option for any of the above described reason a new strategy have been developed in the last decades, the so called interval debulking surgery (IDS). This pathway consists on the anticipation of platinum-based chemotherapy (neoadjuvant treatment) followed by radical surgery and subsequent completion of residual chemotherapy. At present it represents an opportunity to increase the rate of women who could benefit of a cytoreductive surgery.
In this review we explore the current literature and report the evidence about the adoption of IDS, focusing on patients’ selection and the modality of this process.
Resectability criteria
Surgery with cytoreduction intent in advanced ovarian cancer consists of several abdominal procedures to obtain no macroscopic residual disease. For this reason, patients should be referred to gynecologic oncology centers to have access to a dedicated treatment including radical procedures, such as peritonectomy, splenectomy, diaphragmatic stripping or resection, partial liver resection, resection of porta hepatic lesions or distal pancreatectomy (13).
Several studies have shown an increase of complete resection when performing upper abdominal procedures in debulking surgery, thus becoming crucial steps of the cytoreduction (14,15).
However, there are few cases where tumor is considered per se unresectable. Here we report the most common conditions:
- Diffuse carcinomatosis of small bowel;
- Diffuse deep involvement of small bowel mesentery;
- Diffuse infiltration of stomach or duodenum;
- nvolvement of the head or large part of pancreas;
- Multiple hepatic metastasis (multisegmental);
- Multiple lung metastasis;
- Not resectable lymph node disease (e.g., thoracic);
- Brain metastasis.
Operability criteria
Patients should undergo an appropriate work-up to estimate their performance and nutritional status and to define their comorbidities. This step is pivotal to predict the tolerance for an extensive surgery.
Aletti et al. identified patients at higher risk of perioperative complications and who might not benefit of upfront surgery, based on four factors: (I) tumor spread (tumor distribution including stage IV); (II) age (>75 years); (III) performance status [American Society of Anesthesiologists’ (ASA) classification ≥3]; (IV) preoperative albumin (≤3.0 g/dL). The combination of these factors was correlated with too high surgical risks which are not balanced with benefits from aggressive debulking (16).
IDS: current evidence
Even if PDS still represents the standard of care in case of advanced ovarian cancer today, as early as the 1990s some authors claimed a reduced benefit from primary “optimal” cytoreduction in women with a large burden of disease and poor performance status (17,18). Meanwhile, Vergote et al. introduced a new treatment model depending on the extent of the disease and the performance status, the so-called IDS. The preliminary analysis of the above-mentioned authors did not find a detrimental impact of the introduction of this pathway in terms of OS despite a reduction in the rate of PDS from 82% to 57% (18,19).
In 2007, Bristow et al. published a review of 26 non-randomized studies that highlighted the inferiority of NACT compared to PDS in terms of OS, but these were mostly findings based on highly selected data and with a high risk of intrinsic confounders, including both selection and referral biases (20). The first randomized clinical trials that demonstrated noninferiority of NACT + IDS compared to PDS were in fact EORTC55971, published in 2010, and CHORUS, published in 2015 (Table 1).
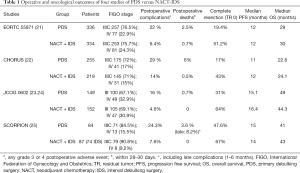
Full table
The EORTC included patients with tubo-ovarian or primary peritoneal cancer stage IIIC–IV and randomized them to receive PDS + adjuvant therapy for six cycles versus three cycles of NACT + IDS. Despite the rate of complete tumor resection was 19.4% in patients in the PDS arm, and 51.2% in the NACT arm, the results demonstrated similar OS of NACT + IDS compared to PDS (30 vs. 29 months respectively). The authors also concluded that the standard of care for women with stage IIIB or earlier stages—a group with a better prognosis than the study population—remained primary cytoreductive surgery but those patients with proven stage IIIC or IV disease might be considered for neoadjuvant chemotherapy (21).
Subsequently, a retrospective analysis of the EORTC55971 attempted to distinguish, within the study population, four subgroups based on two conditions: (I) the clinical stage, (II) the maximum size of the metastatic tumor locations. Based on these criteria, the authors identified two groups of patients who could clearly benefit from one type of treatment or the other: patients with FIGO stage IIIC and maximum metastatic location size <45 mm would have better survival after PDS, patients with stage FIGO IV and maximum size of metastatic locations >45 mm would have had better survival after NACT + IDS (26).
Later in time, the Chemotherapy or Upfront surgery (CHORUS) trial, included patients with tubo-ovarian or primary peritoneal cancer stage III–IV and randomized them to receive PDS + adjuvant therapy for six courses versus three-four courses of NACT + IDS. This trial confirmed the non-inferiority of NACT + IDS compared to PDS in terms of OS (24.1 vs. 22.6 months respectively) (22).Again, the rate of complete tumor resection was lower (17%) in patients in PDS arm than in the NACT arm (39%). In both trials the extremely low rate of complete resection with no residual disease in PDS arms was correlated to the poor survival outcomes, instead the OS of NACT arms were similar to those reported in previous studies.
One of the main advantages of NACT followed by IDS is a reasonable reduction in surgical difficulty and a decrease of postoperative complications, compared with PDS. These benefits were further confirmed in patients with poor performance status. Afterwards, in 2016 two randomized clinical trials explored the perioperative morbidity and mortality in patients treated with PDS versus NACT + IDS (Table 1). The JCOG0602, randomized 301 patients to compare the surgical morbidity. The authors showed that the NACT arm required less radical surgery, shorter operative time and lower rate of abdominal organ and distant metastases resection. Moreover, in the NACT arm they found advantages in terms of blood/ascites loss, albumin transfusion, and severe adverse events after surgery (15.6% vs. 4.6%; P=0.003); as a consequence, authors concluded that NACT treatment was less invasive than PDS and could become the new standard of treatment for advanced ovarian cancer (23). However, the recent oncological results of this trial did not confirm the noninferiority of NACT in terms of survival, the median OS was 49 and 44.3 months in the PDS and NACT groups, respectively (24).
The SCORPION trial randomized 171 patients to PDS versus NACT + IDS to evaluate postoperative complications, PFS, OS and quality of life (QoL) in patients with very high tumor load assessed by a standardized laparoscopic predictive index. Authors found that perioperative moderate/severe morbidity as well as QoL scores were favorable in NACT + IDS arm, probably related to less complex surgery (27). However, NACT did not show any survival advantage compared to PDS despite the selection of patients with high tumor burden (25).
Therefore, it is reasonable to state that NACT + IDS might be a favorable approach in selected cases. First, NACT should reduce the size of the tumor burden to obtain complete cytoreduction more easily and might be an opportunity to optimise the patients prior to IDS, increasing the performance status especially in elderly patients (28). Particularly, a recent pooled data analysis of both EORTC55971 and CHORUS trials demonstrated a statistically significant advantage in progression-free survival and OS with NACT and IDS compared with PDS in the subgroup of patients diagnosed with stage IV disease at presentation, high tumour burden (largest metastatic tumor higher than 5 cm) and poor performance status (29).
Of note, a meta-analysis carried out by Bristow et al. founded that each increase in pre-operative chemotherapy cycles was associated with a decrease in median survival time of 4.1 months, probably related to the development of chemoresistance (30). In this regard, even some retrospective data provided by Bogani et al., suggest that the ideal timing for IDS should be after three cycles of NACT since delaying the IDS to four cycles could worsen OS (31). Moreover, a recent multi-institutional retrospective review analyzing patients who underwent a “delayed” IDS after five or more cycles of chemotherapy demonstrate survival benefit only if a complete resection is achieved (32).
Who and how: the role of laparoscopy
As previously reported, the majority of women with epithelial ovarian cancer (75%) are diagnosed when their disease is already at an advanced stage. So, considering the different rates of complete cytoreduction, postoperative morbidity and perioperative mortality of PDS versus NACT + IDS, it is crucial to identify a tool to predict which patients would benefit from one type of treatment or the other.
Several studies have been performed to find predictors of complete or optimal cytoreduction following PDS. Among these, current non-invasive diagnostic methods including physical examination, ultrasonography, abdominal computed tomography, and serum tumor markers like CA125 and carcinoembryonic antigen were found to be associated with a relatively poor accuracy (33-36). On the other hand, staging laparotomy is probably the most accurate way to determine if the tumor load in the abdomen is too extensive to achieve a complete macroscopic resection; however, this method requires an open approach, which could be high invasive intervention for diagnostic purposes only.
In this scenario, a diagnostic laparoscopy prior to surgery seems to be a valid instrument to assess an accurate prediction of optimal cytoreduction via a minimally invasive approach (37). However, in the literature a huge variability is reported in resectability rates following diagnostic laparoscopy: Vergote et al. evidenced that diagnostic laparoscopy contributed to select patients for primary surgery giving optimal cytoreductive surgery (TR <0.5)in 79% of cases (18); Angioli et al. reported complete cytoreductive surgery (TR 0) in 96% of patients, whose diagnostic laparoscopy showed tumor resectability (38); Fagotti et al. reported optimal cytoreduction (TR ≤1 cm) in only 61% of patients (39). Probably these differences in resectability rates depend on the low reproducibility of the used criteria, which might be driven by subjective individual evaluations, often depending of surgeon’s expertise and own judgement.
In 2008 Fagotti et al. proposed a laparoscopic score based on the presence/absence of omental cake, peritoneal and diaphragmatic extensive carcinosis, mesenteric retraction, bowel and stomach infiltration, spleen and/or liver superficial metastasis. A comparison between laparoscopic and laparotomic evaluation was performed in order to estimate the positive predictive value (PPV), negative predictive value (NPV), and accuracy rate for each parameter. The overall accuracy rate of the laparoscopic procedure ranged between 77.3%, in the case of bowel infiltration, and 100% for peritoneal carcinosis. They assigned to each item an index value of 2, so in the final model, a predictive index score ≥8 identified patients undergoing suboptimal surgery with a specificity of 100% (40). The score was first validated by Brun et al.; authors also proposed their simplified laparoscopy-based score that resulted at least as accurate as the Fagotti score (41).
Laparoscopy may fail to adequately evaluate all patients with advanced stages of ovarian cancer, for example adhesions may impinge the inspection of the entire abdominal cavity and some abdominal regions (the retrohepatic area, the tendinous part of the diaphragm, the suprahepatic veins or the retroperitoneal space) are difficult to assess. However, the data reported above would seem to corroborate the hypothesis that the limit in the evaluation of cytoreducibility of ovarian cancer during a laparoscopy would not be represented so much by the method itself, but rather by the absence of an objective, systematic and reproducible evaluation.
Few studies and a metanalysis have showed the feasibility and safety of complete cytoreductive surgery via minimally invasive surgery in selected ovarian cancer patients with complete/partial response to neoadjuvant chemotherapy (42,43). However, in absence of randomized trial and data about the oncological safety, this approach should be carefully considered in this scenario out of the diagnostic purpose.
Conclusions
NACT represent an effective option for OC treatment; however, at present, optimal management of patients with advanced stage of disease is still debated. The current evidence and guidelines support PDS when feasible. NACT did not show any survival advantage compared to PDS even in selected patients with high tumor load and remains an alternative valid strategy in frail patients and when complete resection might not be achieved in the upfront surgery. The main advantages of this approach are the reduction of perioperative morbidity and mortality and the improvement of QoL. NACT should be not considered as an excuse to avoid highly complex procedures; for this reason, patients should be referred to dedicated gynecologic oncology centers with high expertise in radical surgery.
The ongoing Trial on Radical Upfront Surgery in Advanced Ovarian Cancer (TRUST) has involved gynecologic cancer centers with at least 50% complete resection rate in upfront surgery (44). In a different way of the previous trials, TRUST is focused on patients in whom it was possible to achieve a complete resection with high surgical skills. We hope the results of this trial could help in answering the open questions on this topic.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Federico Ferrari) for the series “Surgical Approaches for Gynecologic Cancers” published in Gynecology and Pelvic Medicine. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://gpm.amegroups.org/article/view/10.21037/gpm-20-61/coif). The series “Surgical Approaches for Gynecologic Cancers” was commissioned by the editorial office without any funding or sponsorship. GB serves as an unpaid editorial board member of Gynecology and Pelvic Medicine from Sept 2019 to Aug 2021. HSM serves as an unpaid editorial board member of Gynecology and Pelvic Medicine from Jun 2020 to May 2022. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Torre LA, Trabert B, DeSantis CE, et al. Ovarian cancer statistics, 2018. CA Cancer J Clin 2018;68:284-96. [Crossref] [PubMed]
- Sant M, Chirlaque Lopez MD, Agresti R, et al. Survival of women with cancers of breast and genital organs in Europe 1999-2007: Results of the EUROCARE-5 study. Eur J Cancer 2015;51:2191-205. [Crossref] [PubMed]
- Lawrie TA, Winter-Roach BA, Heus P, et al. Adjuvant (post-surgery) chemotherapy for early stage epithelial ovarian cancer. Cochrane Database Syst Rev 2015;2015:CD004706 [Crossref] [PubMed]
- Onda T, Yoshikawa H, Yasugi T, et al. The optimal debulking after neoadjuvant chemotherapy in ovarian cancer: proposal based on interval look during upfront surgery setting treatment. Jpn J Clin Oncol 2010;40:36-41. [Crossref] [PubMed]
- Griffiths CT. Surgical resection of tumor bulk in the primary treatment of ovarian carcinoma. Natl Cancer Inst Monogr 1975;42:101-4. [PubMed]
- du Bois A, Reuss A, Pujade-Lauraine E, et al. Role of surgical outcome as prognostic factor in advanced epithelial ovarian cancer: a combined exploratory analysis of 3 prospectively randomized phase 3 multicenter trials: by the Arbeitsgemeinschaft Gynaekologische Onkologie Studiengruppe Ovarialkarzinom (AGO-OVAR) and the Groupe d'Investigateurs Nationaux Pour les Etudes des Cancers de l'Ovaire (GINECO). Cancer 2009;115:1234-44. [Crossref] [PubMed]
- Thigpen T, duBois A, McAlpine J, et al. First-line therapy in ovarian cancer trials. Int J Gynecol Cancer 2011;21:756-62. [Crossref] [PubMed]
- Kobal B, Noventa M, Cvjeticanin B, et al. Primary debulking surgery versus primary neoadjuvant chemotherapy for high grade advanced stage ovarian cancer: comparison of survivals. Radiol Oncol 2018;52:307-19. [Crossref] [PubMed]
- Vergote I, Tropé CG, Amant F, et al. Neoadjuvant chemotherapy is the better treatment option in some patients with stage IIIc to IV ovarian cancer. J Clin Oncol 2011;29:4076-8. [Crossref] [PubMed]
- Chi DS, Musa F, Dao F, et al. An analysis of patients with bulky advanced stage ovarian, tubal, and peritoneal carcinoma treated with primary debulking surgery (PDS) during an identical time period as the randomized EORTC-NCIC trial of PDS vs neoadjuvant chemotherapy (NACT). Gynecol Oncol 2012;124:10-4. [Crossref] [PubMed]
- Kehoe S, Powell J, Wilson S, et al. The influence of the operating surgeon's specialisation on patient survival in ovarian carcinoma. Br J Cancer 1994;70:1014-7. [Crossref] [PubMed]
- Vergote I, Amant F, Kristensen G, et al. Primary surgery or neoadjuvant chemotherapy followed by interval debulking surgery in advanced ovarian cancer. Eur J Cancer 2011;47:S88-S92. [Crossref] [PubMed]
- Colombo N, Sessa C, du Bois A, et al. ESMO-ESGO consensus conference recommendations on ovarian cancer: pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Ann Oncol 2019;30:672-705. [Crossref] [PubMed]
- Papadia A, Morotti M. Diaphragmatic surgery during cytoreduction for primary or recurrent epithelial ovarian cancer: a review of the literature. Arch Gynecol Obstet 2013;287:733-41. [Crossref] [PubMed]
- Pinelli C, Morotti M, Casarin J, et al. The Feasibility of Cardiophrenic Lymphnode Assessment and Removal in Patients Requiring Diaphragmatic Resection During Interval Debulking Surgery for Ovarian Cancer. J Invest Surg 2019; [Crossref] [PubMed]
- Aletti GD, Eisenhauer EL, Santillan A, et al. Identification of patient groups at highest risk from traditional approach to ovarian cancer treatment. Gynecol Oncol 2011;120:23-8. [Crossref] [PubMed]
- Markman M, Hoskins W. Responses to salvage chemotherapy in ovarian cancer: a critical need for precise definitions of the treated population. J Clin Oncol 1992;10:513-4. [Crossref] [PubMed]
- Vergote I, De Wever I, Tjalma W, et al. Neoadjuvant chemotherapy or primary debulking surgery in advanced ovarian carcinoma: a retrospective analysis of 285 patients. Gynecol Oncol 1998;71:431-6. [Crossref] [PubMed]
- Vergote IB, De Wever I, Decloedt J, et al. Neoadjuvant chemotherapy versus primary debulking surgery in advanced ovarian cancer. Semin Oncol 2000;27:31-6. [PubMed]
- Bristow RE, Eisenhauer EL, Santillan A, et al. Delaying the primary surgical effort for advanced ovarian cancer: a systematic review of neoadjuvant chemotherapy and interval cytoreduction. Gynecol Oncol 2007;104:480-90. [Crossref] [PubMed]
- Vergote I, Tropé CG, Amant F, et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med 2010;363:943-53. [Crossref] [PubMed]
- Kehoe S, Hook J, Nankivell M, et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open-label, randomised, controlled, non-inferiority trial. Lancet 2015;386:249-57. [Crossref] [PubMed]
- Onda T, Satoh T, Saito T, et al. Comparison of treatment invasiveness between upfront debulking surgery versus interval debulking surgery following neoadjuvant chemotherapy for stage III/IV ovarian, tubal, and peritoneal cancers in a phase III randomised trial: Japan Clinical Oncology Group Study JCOG0602. Eur J Cancer 2016;64:22-31. [Crossref] [PubMed]
- Onda T, Satoh T, Ogawa G, et al. Comparison of survival between primary debulking surgery and neoadjuvant chemotherapy for stage III/IV ovarian, tubal and peritoneal cancers in phase III randomised trial. Eur J Cancer 2020;130:114-25. [Crossref] [PubMed]
- Fagotti A, Ferrandina MG, Vizzielli G, et al. Randomized trial of primary debulking surgery versus neoadjuvant chemotherapy for advanced epithelial ovarian cancer (SCORPION-NCT01461850). Int J Gynecol Cancer 2020;30:1657-64. [Crossref] [PubMed]
- van Meurs HS, Tajik P, Hof MH, et al. Which patients benefit most from primary surgery or neoadjuvant chemotherapy in stage IIIC or IV ovarian cancer? An exploratory analysis of the European Organisation for Research and Treatment of Cancer 55971 randomised trial. Eur J Cancer 2013;49:3191-201. [Crossref] [PubMed]
- Fagotti A, Ferrandina G, Vizzielli G, et al. Phase III randomised clinical trial comparing primary surgery versus neoadjuvant chemotherapy in advanced epithelial ovarian cancer with high tumour load (SCORPION trial): Final analysis of peri-operative outcome. Eur J Cancer 2016;59:22-33. [Crossref] [PubMed]
- Pinelli C, Morotti M, Casarin J, et al. Interval Debulking Surgery for Advanced Ovarian Cancer in Elderly Patients (≥70 y): Does the Age Matter? J Invest Surg 2020; [Crossref] [PubMed]
- Vergote I, Coens C, Nankivell M, et al. Neoadjuvant chemotherapy versus debulking surgery in advanced tubo-ovarian cancers: pooled analysis of individual patient data from the EORTC 55971 and CHORUS trials. Lancet Oncol 2018;19:1680-7. [Crossref] [PubMed]
- Bristow RE, Chi DS. Platinum-based neoadjuvant chemotherapy and interval surgical cytoreduction for advanced ovarian cancer: a meta-analysis. Gynecol Oncol 2006;103:1070-6. [Crossref] [PubMed]
- Bogani G, Matteucci L, Tamberi S, et al. The Impact of Number of Cycles of Neoadjuvant Chemotherapy on Survival of Patients Undergoing Interval Debulking Surgery for Stage IIIC-IV Unresectable Ovarian Cancer: Results From a Multi-Institutional Study. Int J Gynecol Cancer 2017;27:1856-62. [Crossref] [PubMed]
- Plett H, Filippova OT, Garbi A, et al. Role of delayed interval debulking for persistent residual disease after more than 5 cycles of chemotherapy for primary advanced ovarian cancer. An international multicenter study. Gynecol Oncol 2020;159:434-41. [Crossref] [PubMed]
- Chi DS, Venkatraman ES, Masson V, et al. The ability of preoperative serum CA-125 to predict optimal primary tumor cytoreduction in stage III epithelial ovarian carcinoma. Gynecol Oncol 2000;77:227-31. [Crossref] [PubMed]
- Nelson BE, Rosenfield AT, Schwartz PE. Preoperative abdominopelvic computed 549 tomographic prediction of optimal cytoreduction in epithelial ovarian carcinoma. J Clin Oncol 1993;11:166-72. [Crossref] [PubMed]
- Dowdy SC, Mullany SA, Brandt KR, et al. The utility of computed 555 tomography scans in predicting suboptimal cytoreductive surgery in women with 556 advanced ovarian carcinoma. Cancer 2004;101:346-52. [Crossref] [PubMed]
- Rutten MJ, van de Vrie R, Bruining A, et al. Predicting surgical outcome in patients with International Federation of Gynecology and Obstetrics stage III or IV ovarian cancer using computed tomography: a systematic review of prediction models Int J Gynecol Cancer 2015;25:407-15. [Crossref] [PubMed]
- Rutten MJ, Leeflang MM, Kenter GG, et al. Laparoscopy for diagnosing resectability of disease in patients with advanced ovarian cancer. Cochrane Database Syst Rev 2014;2014:CD009786 [Crossref] [PubMed]
- Angioli R, Palaia I, Zullo MA, et al. Diagnostic open laparoscopy in the management of advanced ovarian cancer. Gynecol Oncol 2006;100:455-61. [Crossref] [PubMed]
- Fagotti A, Fanfani F, Ludovisi M, et al. Role of laparoscopy to assess the chance of optimal cytoreductive surgery in advanced ovarian cancer: a pilot study. Gynecol Oncol 2005;96:729-35. [Crossref] [PubMed]
- Fagotti A, Ferrandina G, Fanfani F, et al. Prospective validation of a laparoscopic predictive model for optimal cytoreduction in advanced ovarian carcinoma. Am J Obstet Gynecol 2008;199:642.e1-642.e6. [Crossref] [PubMed]
- Brun JL, Rouzier R, Uzan S, et al. External validation of a laparoscopic-based score to evaluate resectability of advanced ovarian cancers: clues for a simplified score. Gynecol Oncol 2008;110:354-9. [Crossref] [PubMed]
- Cardenas-Goicoechea J, Wang Y, McGorray S, et al. Minimally invasive interval cytoreductive surgery in ovarian cancer: systematic review and meta-analysis. J Robot Surg 2019;13:23-33. [Crossref] [PubMed]
- Brown J, Drury L, Crane EK, et al. When Less Is More: Minimally Invasive Surgery Compared with Laparotomy for Interval Debulking After Neoadjuvant Chemotherapy in Women with Advanced Ovarian Cancer. J Minim Invasive Gynecol 2019;26:902-9. [Crossref] [PubMed]
- Reuss A, du Bois A, Harter P, et al. TRUST: Trial of Radical Upfront Surgical Therapy in advanced ovarian cancer (ENGOT ov33/AGO-OVAR OP7). Int J Gynecol Cancer 2019;29:1327-31. [Crossref] [PubMed]
Cite this article as: Pinelli C, Guerrisi R, Brusadelli C, Artuso V, Majd HS, Bogani G, Ghezzi F, Casarin J. Interval debulking surgery for advanced ovarian cancer: when, how and why? Gynecol Pelvic Med 2021;4:16.


